 The Phase IIb clinical trial involving only adult subjects was conducted in North America (the United States and Canada), Europe (France, Germany, and Poland), and Australia. The MWIC of the SD NRS had not been established prior to this study and therefore needed to be established from the target population using multiple data sources collected at the patient level (phase II data, and qualitative interviews). To address our objective, we used an explanatory sequential MMR design using different study samples. Our work aims to establish MWIC thresholds of a new PRO, a sleep disturbance numerical rating scale (SD NRS ©), in adults and adolescents with moderate-to-severe atopic dermatitis (AD), using a mixed method approach in the context of a drug development program. Furthermore, the US regulators do recognize that emerging approaches such as MMR may be used to triangulate and interpret COA-based endpoint results. The US Food and Drug Administration (FDA) has even recommended MMR as a research methodology to collect comprehensive patient community input on the burden of disease and current therapy as well as to identify what is important to patients. In the field of health outcomes research, MMR is now well accepted and commonly used. MMR has been well established for more than 50 years in the social behavioral sciences. MMR is defined as “a research in which the investigator collects and analyses data, integrates the findings, and draws inferences using both qualitative and quantitative approaches or methods in a single study or program of inquiry”. This includes mixed-methods research (MMR), combining quantitative methods using data on change scores from clinical trials or observational studies and qualitative methods using data from concept elicitation, cognitive interviewing, or exit interviews. Over the last few years, there has been a growing interest in developing novel methods for establishing thresholds for meaningful change of scores such as the use of semi-structured interviews, vignettes (standard setting), surveys, conjoint analysis, and Delphi panels. These traditional approaches for interpreting COA scores have both advantages and disadvantages that have been discussed elsewhere. Establishing meaningful change of a COA score is commonly assessed using anchor-based and distribution-based methods. Meaningful change can also be interpreted at the group level to evaluate clinically important differences (CID) between two clinically distinct groups (e.g., experimental treatment and placebo) that have meaningful differences in their level of change in scores. Interpretation of meaningful change at the individual level (i.e., meaningful within-individual change or responder definition) corresponds to the amount of change in scores reported by any individual over a predetermined time period that should be interpreted as a benefit to patients. It includes examination of thresholds for within-individual change as well as between-group differences. This mixed-methods design provides interesting insights for establishing MWIC thresholds of a PRO score in the context of a drug development program.Įstablishing what constitutes a meaningful change score of clinical outcome assessments (COAs), including patient-reported outcomes (PROs), is essential for interpreting results based on COA endpoints used for evaluating treatment benefit. This research determined MWIC threshold estimates for the SD NRS score in both adolescents and adults with moderate-to-severe AD using an explanatory sequential mixed-methods design. Triangulation of results from both quantitative and qualitative approaches suggested that a 2- to 6-point decrease in the SD NRS score change constitutes a preliminary range of MWIC threshold estimates. Quantitative anchor-based and distribution-based approaches supported by qualitative-based approaches were conducted, and results were triangulated to determine preliminary MWIC thresholds of the SD NRS score. This mixed-methods design used phase IIb data and a stand-alone qualitative study. MethodsĪn explanatory sequential mixed-methods design was used to address the research objective. This research aims to establish a MWIC threshold of the SD NRS score in the context of a drug development program. 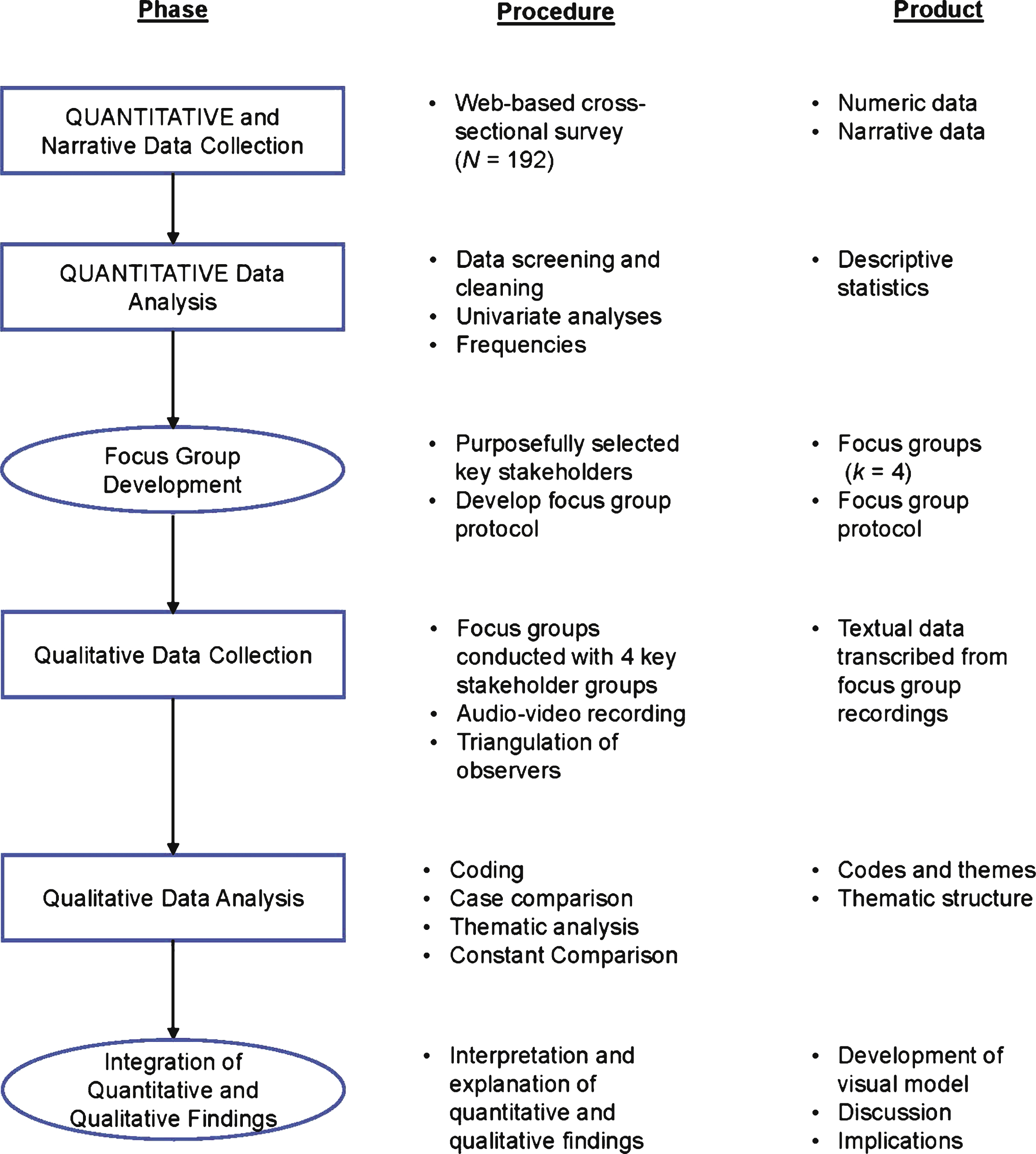 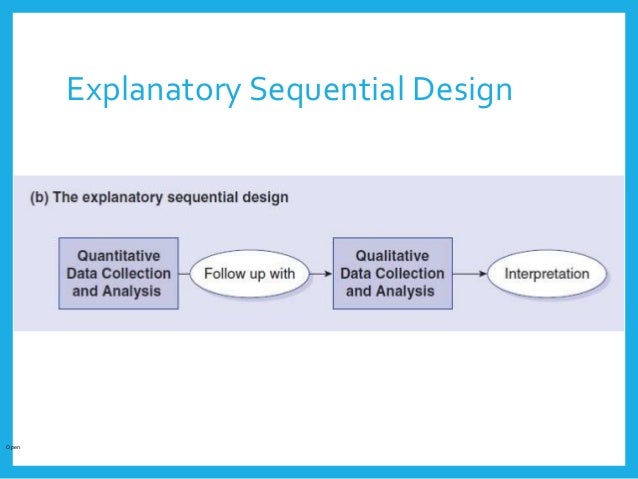
A new patient-reported outcome (PRO), a sleep disturbance numerical rating scale (SD NRS), was developed in adults and adolescents with moderate-to-severe atopic dermatitis (AD). Establishing a meaningful within-individual change (MWIC) threshold is a key aspect for interpreting scores used as endpoints for evaluating treatment benefit. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
